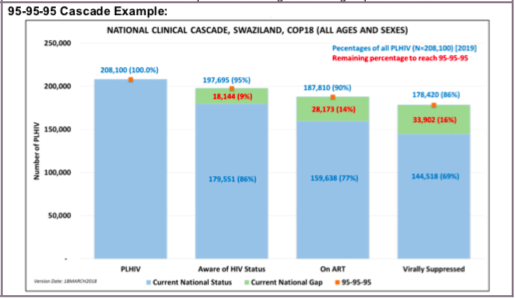
(TX_CURR) Number of adults and children currently receiving antiretroviral therapy (ART)
Export Indicator
This indicator measures the ongoing scale-up and uptake of ART and retention in ART programs as a critical step in the HIV service cascade and assesses progress towards coverage of ART for all eligible HIV-positive individuals when reviewed against the number of PLHIV that are estimated to be eligible for treatment. It allows us to track the response to the epidemic in specific geographic areas and among specific populations as well as at the national level. Disaggregations by age and sex can help better understand which populations are at epidemic control and which populations are lagging behind. Lastly, newly added disaggregations on ARV dispensing quantity can be used to determine uptake of MMD at PEPFAR sites, in PEPFAR SNUs, and across PEPFAR partners.
Number of adults and children currently receiving antiretroviral therapy (ART)
N/A
How to collect:
This indicator should be collected from facility ART registers/databases, program monitoring tools, and drug supply management systems.
Count the number of adults and children who are currently receiving ART in accordance with the nationally approved treatment protocol (or WHO/UNAIDS standards) at the end of the reporting period. Importantly, patients who have not received ARVs within four weeks (i.e., 28 days) of their last missed drug pick-up should not be counted.
The following should also be considered:
- Patients on ART who initiated or transferred-in during the reporting period should be counted.
- Patients that pick up 3 or more months of anti-retroviral drugs at one visit (i.e., multi-month dispensation) should also be counted as long as they have received enough ARVs to last to the end of the reporting period at a minimum.
- However, if it is determined that a patient has died, they should immediately be removed from the TX_CURR results.
- HIV-positive pregnant women who are eligible for and are receiving antiretroviral drugs for their own treatment should be counted. HIV-positive pregnant women initiating lifelong ART through PMTCT (Option B+) will count as “current” on ART under this indicator. These include HIV-infected pregnant women who:
- Have newly initiated ART during the current pregnancy
- Are already on ART at the beginning of the current pregnancy
Patients excluded from the current on ART count are patients who died, stopped treatment, transferred out, or are lost to follow-up. Patients who have not received ARVs within four weeks (i.e., 28 days) of their last missed drug pick-up should not be counted. Patients do not need to qualify as lost to follow-up before tracing efforts commence. Efforts to trace patients that have missed a clinical visit or drug pick-up should begin immediately following a missed clinical contact.
Patients who have not received ARVs within four weeks of their last missed drug pick-up should be described further in the reporting of the TX_ML indicator. Patients that restart treatment after four weeks or more of being off ARVs should also be counted under TX_RTT in the reporting period in which the patient returns to care and restarts ARVs.
TX_CURR should be reported from both PEPFAR-supported sites in the private and/or public sector. Patients currently receiving treatment from mobile clinics can be reported in two ways. Firstly, if the mobile clinic is associated with (e.g., receives commodities, reports to, is staffed by) a nearby health facility, then these individuals should be reported by that facility. Secondly, if a mobile clinic is stationary for more than 2 reporting periods, it should be added to the PEPFAR facility list with geocodes and data should be reported for this mobile clinic directly.
DO NOT include: Patients who receive ARVs for post-exposure prophylaxis (PEP) or short-term ART only for prevention (PrEP) should not be reported in this indicator.
Key Populations (KPs):
Both KP-specific and clinical partners should complete these KP disaggregations, but only if safe to maintain these files and to report. Reporting of the key population disaggregation should be consistent with what is described under the KP_PREV “How to review for data quality” section on mutual exclusivity of an individual who falls under multiple KP categories (e.g., FSW who injects drugs). In such instances, the individual should only be reported in ONE KP disaggregation category with which this person is most identified. See Appendix A to support the identification of key populations at service delivery.
The first priority of data collection and reporting of treatment among key populations must be to do no harm. These data must be managed confidentially to ensure the identities of individuals are protected and to prevent further stigma and discrimination of key populations.
Reporting level: Facility
How to review for data quality:
- Confirm that TX_CURR ≥ TX_NEW
- Confirm that TX_CURR ≥ TX_RTT
- Confirm that TX_CURR ≥ Disaggregates for ARV Dispensing Quantity
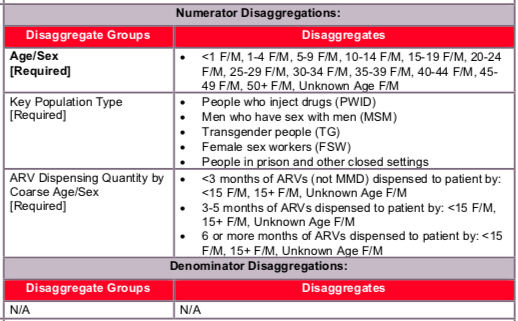
Disaggregate descriptions & definitions:
For age/sex disaggregates:
CURRENT is a state defined by treatment status when last seen, so it is expected that characteristics of these clients would be updated each time they are seen by a program. Age represents an individual’s age at the end of the reporting period or when last seen at the facility. For example, a 14-year-old child will be counted as currently receiving treatment in the <15 age category at the end of reporting period “A”. During reporting period “B” the child turns age 15 and so at the end of this reporting period the child will be counted under the 15+ age category.
For ARV dispensing quantity by coarse age/sex disaggregates:
Patients should be categorized by the coarse age disaggregates while being further categorized by the months of ARVs dispensed: <3 months of ARVs dispense to the patient, 3-5 months of ARVs dispensed to the patient, or 6 or more months of ARVs dispensed to the patient. By definition, patients dispensed just one or two month of ARVs are not receiving MMD. However to ensure data completeness and quality, they are collected herein.
NOTE: MMD should not be confused with multi-month prescriptions. For example, patients receiving 6-month prescriptions that the facility fulfills in two refills of a 3 month supply can be counted as receiving MMD in the 3-5 month MMD disaggregate. Inversely, patients receiving 6-month prescriptions that the facility fulfills in six refills of a 1 month supply would be counted in the <3 months disaggregated and would not be considered as receiving MMD.
Count the number of adults and children who are currently receiving ART.
Indicator changes (MER 2.0 v2.3 to v2.4):
- New disaggregation to collect the quantity of ARVs dispensed to each patient was added to monitor uptake of multi-month dispending (MMD).
- Key populations disaggregations added in order to ensure appropriate monitoring of the full HIV clinical cascade across key populations.
PEPFAR-support definition:
Standard definition of DSD and TA-SDI used.
Provision of key staff or commodities for PLHIV receiving ART includes: the provision of key staff and/or commodities can include ongoing procurement of critical commodities, such as ARVs, or funding for salaries of HCW who deliver HIV treatment services. Staff who are responsible for the completeness and quality of routine patient records (paper or electronic) can be counted here; however, staff who exclusively fulfill MOH and donor reporting requirements cannot be counted.
Ongoing support for PLHIV receiving ART service delivery improvement includes: clinical mentoring and supportive supervision of staff at HIV sites providing ART, support for quality improvement activities, patient tracking system support, routine support of ART M&E and reporting, commodities consumption forecasting and supply management
- What percentage of clients are picking up their ART drugs on a quarterly basis? On a semi-annual basis?
- What percentage of clients are being seen for clinical follow-up visits on a quarterly basis? On a semi-annual basis? On an annual basis?
- Describe differences in MMD uptake across age and sex groups and sites/SNUs.