(TB_PREV) Proportion of ART patients who started on a standard course of TB Preventive Treatment (TPT) in the previous reporting period who completed therapy
Export Indicator
This indicator measures the performance of HIV programs in scaling up TPT, with the goal of preventing progression to active TB disease among PLHIV and decreasing ongoing TB transmission in this population.
As part of a cascade from TX_CURR to TB screening (captured in TX_TB), this indicator will inform programs on the pace of scale-up, and the proportion will allow for monitoring of cohorts through completion of therapy.
Disaggregates on the timing of ART and age/sex breakdowns will allow programs to monitor those who are newly starting ART, an important focal population in all countries and in particular in countries that have already provided TPT for many of their PLHIV in care.
Among those who started a course of TPT in the previous reporting period, the number that completed a full course of therapy (for continuous IPT programs, this includes the patients who have completed the first 6 months of isoniazid preventive therapy (IPT), or any other standard course of TPT such as 3 months of weekly isoniazid and rifapentine, or 3-HP)
Number of ART patients who were initiated on any course of TPT during the previous reporting period
How to calculate annual total:
The TB_PREV denominator and numerator should be analyzed independently of other data and the results reported in Q2 and Q4 should be summed to calculate the total number of ART patients who initiated and completed a course of TPT.
When analyzing this data in conjunction with data on TB screening for ART patients (TX_TB), it is important to align the correct reporting periods. For example, TB_PREV captures those who were initiated on TPT during the previous reporting period, so it should be compared to TB screening (TX_TB Denominator) and TX_CURR data from the previous reporting period.
How to collect:
The denominator can be generated by counting the total number of patients who initiated any regimen of TPT in the semiannual reporting period that is prior to the one being reported on. For example, if reporting is for Q1 and Q2 of a fiscal year (e.g., October 2019 to March 2020), then the denominator would include those that were started on TPT in Q3 and Q4 of the previous fiscal year (e.g., April to September 2019). If a TPT register is being used, then this would require simply framing out the dates that define the previous reporting period and counting all those who started TPT.
Importantly, programs should ensure that patients on continuous isoniazid therapy are counted only once, when they initiate therapy (denominator) and after they complete the first six months (numerator); care should be taken to ensure they are not included in future calculations.
If a patient is initiated on TPT and dies before TPT completion, this patient should be recorded in the denominator, but not in the numerator. If a patient initiates TPT at one site, completes at another, and is a documented transfer, that patient should be recorded in the numerator at the site where they initiated TPT, and they should be recorded as completed TPT (numerator) at the new site.
The numerator can be generated by counting the subset of patients from the denominator who received at least six months of IPT or have completed another standard course of TPT. If a TPT register is being used, this would require framing out the dates that define the previous reporting period, identifying those that initiated TPT during the reporting period (the denominator) and then documenting the number of those patients who completed the course of TPT that they started during that reporting period. This should include the patients who completed a shorter alternative course, such as 3-HP, as well as those who are on prolonged or continuous IPT who have completed their first six months of therapy.
Note: If a patient was started on IPT in the previous reporting period (e.g., Q3 or Q4 FY2019), he/she would have completed during the current reporting period (e.g., Q1 or Q2 FY2020).
For IPT:
- All patients who started any form of IPT, including prolonged or continuous IPT, at any time in the previous 6-month reporting period (i.e., at any time in the 6 months before the start of the period being reported) should be included in the denominator. Among the denominator, those that completed at least six months of isoniazid therapy would have done so in the period currently being reported (the numerator). The few patients who started and completed IPT in the previous reporting should be included and counted.
For 3-HP:
- Patients who are taking 3-HP may have initiated and completed therapy in the previous reporting period, or they may have initiated TPT in the previous reporting period and completed TPT in the period currently being reported.
- Any patient who started 3-HP at any point in the previous reporting period would be included in the denominator.
Any patient from that denominator who completed the course would be included in the numerator; this would include those who completed 3-HP in the first 3 months of the period being reported on.
For alternative regimens:
- Patients who are taking other regimens (such as 1-HP) may also have initiated and completed therapy in the previous reporting period or they may have initiated TPT in the previous reporting period and completed TPT in the period currently being reported. Include and count patients under both scenarios (start and completion in the same reporting period AND start in the previous reporting period but completion in the one currently being reported).
These data elements can be collected from the ART register or from separate TPT registers. In some countries, TB presumptive registers might contain this information as well, but the information will need to be cross referenced for ART status.
How to review for data quality:
Reporting Level: Facility
Reporting frequency: Semi-Annually
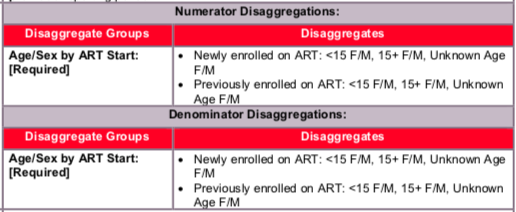
Age/Sex by ART Start Descriptions:
- Newly enrolled on ART: These individuals initiated TPT within 6 months of being enrolled on ART; data to be submitted by the following disaggregates: <15F/M, 15+F/M Unknown Age F/M
- Previously enrolled on ART: These individuals initiated TPT at least 6 months (or longer) after being enrolled on ART; data to be submitted by the following disaggregates: <15F/M, 15+F/M, Unknown Age F/M
The numerator is generated by counting the number of PLHIV on ART from the previous reporting period who were documented as having received at least six months of IPT or having completed any other standard course of TPT (such as 3-HP).
The denominator is generated by counting the total number of patients who were started on ART who were started on any course of TPT during the reporting period prior to the one being reported.
Indicator changes (MER 2.0 v2.3 to v2.4):
- The denominator was changed from the number of ART patients who “are expected to complete a course of TPT” to those who were initiated on any course of TPT during the previous reporting period.
The therapy type (regimen) disaggregates for the numerator and denominator were removed. The previous disaggregates were Age/Sex by Type of TB Preventive Therapy by ART Start, and they are now Age/Sex by ART Start. TPT regimen type should now be captured in the indicator narrative.
A narrative question has been included to capture information on TPT regimen type.
For clarity, the APR calculation for TB_PREV was changed from a snapshot indicator, to being summed over time (i.e., previous calculation: APR=Q4, new calculation: APR=Q2+Q4).
PEPFAR Support definition:
Standard definition of DSD and TA-SDI used.
Provision of key staff or commodities for routine HIV-related services includes: ongoing provision of critical re-occurring costs or commodities (such as ARVs, TB preventive therapy and diagnostic/screening tests) or funding of salaries or provision of Health Care Workers for HIV clinic services. Staff responsible for maintaining patient records in both HIV and TB clinics are included in this category however staff responsible for fulfilling reporting and routine M&E requirements are not included.
Ongoing support for patients receiving routine HIV-related services includes: training of HIV service providers, clinical mentoring and supportive supervision of staff at HIV sites, infrastructure/renovation of facilities, support of HIV service data collection, reporting, data quality, QI/QA of HIV services support, ARV and TPT consumption forecasting and supply management, support of lab clinical.
- What proportion of patients who completed TPT received IPT, 3-HP, or an alternative TPT regimen (e.g., 1-HP)?
Roughly what proportion of patients who received TPT were treated with the 6-month isoniazid regimen?
Broadly describe the main reasons why TPT was not completed (e.g., adverse events, patients were lost to follow up, patients refused to continue, etc.).
Roughly what proportion of all PLHIV on treatment have already completed TB preventive therapy prior to this reporting period (and were not eligible for TPT and not include in this indicator)?
If TB preventive therapy was not provided to all PLHIV in care, what are the main reasons for limited scale-up?
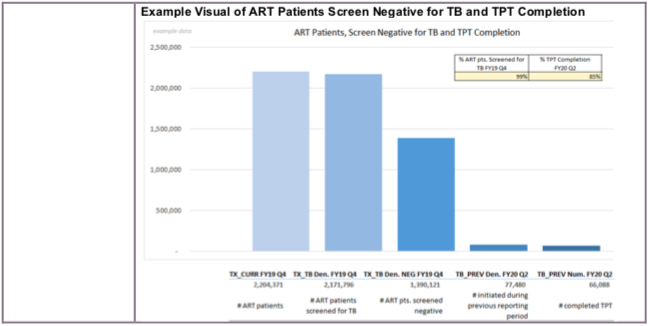
Data Visualization & Use Examples:
*see the next page for visual example*
Related Indicators
TB.2 TPT completion, 2020, WHO Consolidated HIV strategic information guidelines: driving impact through programme monitoring and management (https://www.who.int/publications/i/item/consolidated-hiv-strategic-information-guidelines).