(PMTCT_STAT) (including PMTCT_STAT_POS) Percentage of pregnant women with known HIV status at antenatal care (includes those who already knew their HIV status prior to ANC)
Export Indicator
Track progress toward ensuring that all pregnant women who attend PEPFAR-supported antenatal care (ANC) know their HIV status and those newly testing positive are initiated on ART.
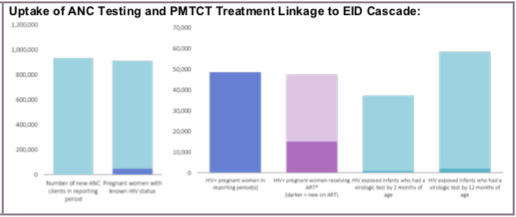
Number of pregnant women with known HIV status at first antenatal care visit (ANC1) (includes those who already knew their HIV status prior to ANC1)
Number of new ANC clients in reporting period
How to collect:
The data source is the ANC register. There is a risk of double counting as a pregnant woman could be tested multiple times during one pregnancy; therefore, partners should ensure a data collection and reporting system is in place to minimize double counting, including a longitudinal ANC register (meaning a register that is able to record all information about one pregnancy in one location, with rows or columns that allow for recording information on multiple visits during that pregnancy).
Subsequent testing during pregnancy and breastfeeding should be counted in the HTS modality: Post ANC1: Pregnancy/L&D/BF. There is also a risk of undercounting if those women who already knew their HIV status prior to attending ANC are not documented, therefore the ANC register should at a minimum document both “previously known positive” and “newly tested positive”.
It may be appropriate to report “known negative” women under the “Recent Negative” disaggregate if national guidelines do not require retesting women known to be HIV negative (often women tested in the last 3 months, however exact timing depends on local guidelines). See disaggregate definitions below for additional information.
Women reported under the “Newly Tested Positive” and “New Negative” disaggregations will auto-populate the HTS_TST ANC1 modality. Women who are tested later in pregnancy, during L&D, and/or during breastfeeding should be reported under the HTS_TST Post ANC1: Pregnancy/L&D/BF modality.
Reporting level: Facility
How to review for data quality:
The % should never be above 100% at a site, and therefore review of the method of data collection and correction of any errors at sites with greater than 100% coverage is important to ensuring data quality for this indicator.
Retesting of HIV-negative women during pregnancy, at L&D, and through the postpartum period is an important program strategy and is collected under the HTS_TST Post ANC1: Pregnancy/L&D/BF modality. Please see the HTS_TST reference sheet for more information on collecting this information.
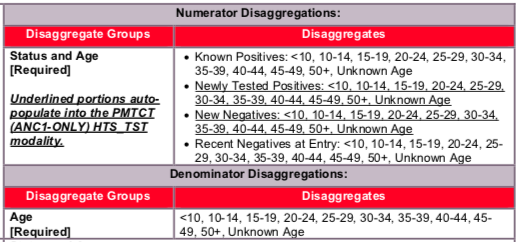
- Known Positive at entry: Number of pregnant women attending ANC for a new pregnancy who were tested and confirmed HIV-positive at any point prior to the current pregnancy should be reported as known positive at entry. Pregnant women with known HIV status attending ANC for a new pregnancy may not need retesting if they are already on ART, or they may be required to be retested prior to initiating ART based on national guidelines. Known positives who are re-tested and confirmed to be HIV positive prior to initiating ART should still be documented as known positive at entry.
- Newly Tested Positive: The number of women attending ANC1 who were tested for HIV and received a positive result. Women who tested negative prior to this pregnancy and are tested again at ANC1 for this new pregnancy should be counted in this indicator.
- New Negatives: The number of women attending ANC1 who were tested for HIV and received a negative result. Women who tested negative prior to this pregnancy and are tested again at ANC1 should be counted in this indicator.
- Recent Negative at entry: Number of pregnant women attending ANC for a new pregnancy who recently tested HIV negative and are not eligible – according to country clinical guidelines - for another HIV test at ANC1. For example, women who tested negative within three months of attending ANC1 may not be recommended for testing per country clinical guidelines. This is expected to be a less utilized disaggregate.
The numerator is the sum of the following two data elements:
- The number of women with a previously known HIV status (both known HIV positive and known negative) attending their first ANC visit (ANC1) for a new pregnancy over the last reporting period.
- The number of women attending ANC1 who were tested for HIV and received results
N/A
Indicator changes (MER 2.0 v2.3 to v2.4): None
PEPFAR Support definition:
Standard definition of DSD and TA-SDI used.
Provision of key staff or commodities for PMTCT includes: commodities such as test kits, ARVs, lab commodities, or funding for salaries of health care workers.
Ongoing support for PMTCT service delivery improvement includes: training of PMTCT service providers, clinical mentoring and supportive supervision of PTMCT service sites, infrastructure/renovation of facilities, support for PMTCT service data collection, reporting, data quality, QI/QA of PMTCT services support, ARV consumption forecasting and supply management, support of lab clinical monitoring of patients, supporting patient follow- up/retention, support of mother mentoring programs.
Guiding narrative questions:
- Provide context for poor performance in PMTCT_STAT coverage (Numerator/Denominator = STAT coverage) by geographic area, age, or partner/implementing mechanism, including any planned activities/remedial actions.
- For areas where age disaggregates are NOT completely reported, describe challenges for collecting and/or plan and timeline for collection.