(PMTCT_FO) Percentage of final outcomes among HIV exposed infants registered in a birth cohort
Export Indicator
In settings where national guidelines support breastfeeding of HIV-exposed infants, antibody testing of all HIV-exposed children at 18 months of age and/or 6 weeks after cessation of breastfeeding is recommended to determine final HIV status (‘final outcome’/FO) of HIV-exposed children. To accomplish this goal, it is recommended to identify infants at birth or at the first infant follow-up visit and track them through the end of the breastfeeding period. This indicator measures progress toward ensuring that all infants born to HIV-positive women have an outcome documented. In settings where a mother- infant register is utilized and/or it is common practice for HIV-infected women to breastfeed less than or more than 18 months please describe in the narrative the final outcome time point.
Number of HIV-exposed infants with a documented outcome by 18 months of age disaggregated by outcome type.
(Note: Collection of 18 month visit outcomes is recommended at 24 months of age, see additional explanation to the right.)
Number of HIV-exposed infants who were born 24 months prior to the reporting period and registered in the birth cohort.
How to collect:
To report on this indicator PEPFAR supported sites would ideally use registers or facility held cards for HIV exposed infants that collect longitudinal information on follow-up and are organized by birth month of infants. This methodology is referred to as birth cohort reporting.
Two examples of birth cohort reporting:
- In Kenya, this indicator was first piloted by PEPFAR and the Ministry of Health in Western Kenya and is currently integrated into the national HIV summary reporting tool. Data from the facility HIV exposed infant longitudinal follow-up register, which organizes infants by birth-month cohorts, are aggregated into a report summarizing outcomes for infants reaching 24 months of age during each month.
- In Malawi, clinic staff complete monthly follow up reporting forms as part of the national quarterly supervision visits using data collected directly from HIV-exposed infant cards which are kept in a binder that is organized by birth month (no HIV exposed register is used).
As an example, for those infants born in FY 2018, the outcomes would be reported in FY 2020.
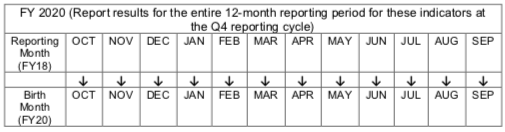
Both approaches allow a paper-based health facility records to quickly identify the number of HIV-exposed infants registered in the birth cohort at any time between 0 and 18 months of age (denominator).
How to review for data quality:
By design this indicator should equal 100% if all outcomes are known regardless of outcome type. This allows for facilities to check that all HIV-exposed infants have an outcome assigned to them during the reporting process. Data utilization requires reviewing the disaggregated data to understand the specific outcomes of interest. In settings where HIV-exposed infant registers do not allow for documentation of all disaggregated outcomes, country teams should report only on available disaggregates even if the aggregate indicator is less than 100%, however this should be specified in the narrative.
The denominator should include those “Transferred In” and those “Transferred Out” as long as for “Transferred In” there is documentation that HIV-exposed infants were registered at their original site in the birth cohort at any time between 0 and 18 months of age and were born 24 months prior to the reporting period. “Transferred Out” should be reported under HIV status unknown. The inclusion of Transfers-In/Out provides a quality check to ensure that all exposed infants have an outcome assigned to them during the reporting process such that the sum of the numerator disaggregation equals the denominator. However, this may lead to outcomes for >100% of HIV positive pregnant women (PMTCT_STAT_POS) identified at a site so this comparison should not be used as a logic check.
Reporting level: Facility

Disaggregate descriptions & definitions:
Outcome type:
For the numerator to be calculated, implementing partners are required to report:
- HIV-infected: Number of HIV-exposed infants identified as HIV-infected at any point during follow-up. HIV-infected includes infants and children with diagnostic virologic or serologic confirmation of HIV-infection (DNA PCR before 18 months; rapid test at 18 months) and those with a presumptive HIV diagnosis where DNA PCR is not available. Site should also maintain data on HIV infected infants and whether they are linked or not linked to ART services, or whether they have no information on patient linkage to ART programs.
- HIV-uninfected: Number of HIV-exposed infants with a negative 18-month antibody test documented. Based on national guidelines, countries should determine if “HIV- uninfected” includes infants with a documented negative antibody test that was done at least 6 weeks after cessation of breastfeeding but before 18 months of age.
- HIV final status unknown: Sum of the following disaggregates (not reported in DATIM but should be documented at site level)
- In care but no test done: Number of HIV-exposed infants who attended 18-month visit but no antibody test result is documented (unknown FO)
- Lost to follow-up: Number of HIV-exposed infants who did not attend the 18-month visit (unknown FO)
- Transferred out (unknown FO): Number of HIV-exposed infants who transferred out between 0 and 18 months without confirmation of HIV-infection (unknown FO)
- Died without status known: Number of HIV-exposed infants who are documented to have died without confirmation of HIV-infection between 0 and 18 months. Note: HIV- exposed infants who are HIV infected and later confirmed to have died or transferred out during follow-up are still counted under HIV infected and not died or transferred out.
Every infant in a given cohort should be assigned one outcome only.
Calculated indicator in DATIM, sum of: HIV- infected, HIV-uninfected, HIV-final status unknown, died without status known.
It is recommended to wait to collect the 18 month visit outcomes until the patient is 24 months old for the following reasons: 1) this allows for children who present several months late to their 18 month visit to be included in the numerator and 2) cohort reporting is easiest when monthly reporting by facilities is used and where the birth month and the reporting month are the same calendar month (i.e., for infants born in January 2012, their 24 month reporting month would be January 2014, rather than using the 18 month reporting month of July 2013).
Only those HIV-exposed infants registered in the birth cohort at any time between 0 and 18 months of age (including transfers-ins) who were born 24 months prior to the reporting period are included in the denominator.
Indicator changes (MER 2.0 v2.3 to v2.4): None
PEPFAR Support definition:
Standard definition of DSD and TA-SDI used.
Provision of key staff or commodities for PMTCT include: commodities such as test kits, ARVs, lab commodities, or funding for salaries of health care workers.
Ongoing support for PMTCT service delivery improvement includes: training of PMTCT service providers, clinical mentoring and supportive supervision of PTMCT service sites, infrastructure/renovation of facilities, support for PMTCT service data collection, reporting, data quality, QI/QA of PMTCT services support, ARV consumption forecasting and supply management, support of lab clinical monitoring of patients, supporting patient follow- up/retention, support of mother mentoring programs.
Guiding narrative questions:
- Provide context for PMTCT_FO results (e.g., PMTCT_FO not equal to 100%, low or high rate of HIV-uninfected infants) and describe how this data being use for program management?
- Provide context on:
- The status of birth cohort monitoring in your operating unit, geographic area or partner/implementing mechanism, including any planned activities.
- The data source used for reporting, and any key information about data quality that is important for interpretation of results (see MER reference sheet for examples).
- The number and proportion of PEPFAR-supported PMTCT sites implementing cohort monitoring and able to (1) report on PMTCT_FO and (2) longitudinally track mothers to assess retention/viral suppression