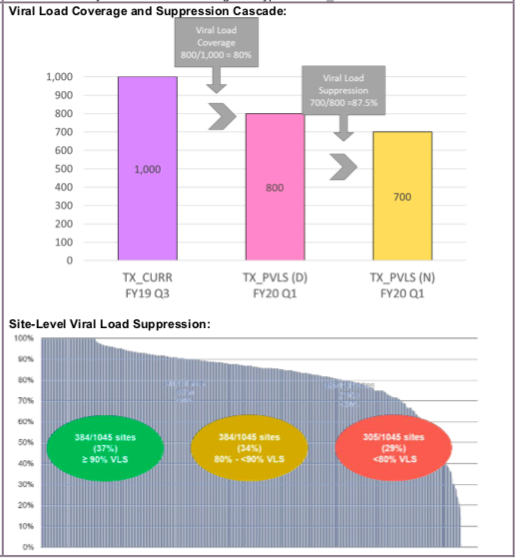
(TX_PVLS) Percentage of ART patients with a suppressed viral load (VL) result (<1000 copies/ml) documented in the medical or laboratory records/laboratory information systems (LIS) within the past 12 months
Export Indicator
VL SUPPRESION OUTCOMES:
This indicator monitors the proportion of documented viral load results from adult and pediatric ART patients who have been on ART for at least 3 months (or according to national guidelines) with a suppressed result (<1,000 copies/ml). This allows ART programs to monitor individual and overall programmatic response to ART as measured by virologic suppression. This indicator will provide data on patients who have a viral load (VL) test in the past 12 months and the percentage who were virally suppressed at the most recent test.
VL TESTING COVERAGE:
Comparison of the denominator for this indicator with the result for TX_CURR from 6 months earlier (i.e., two quarters prior) can be used to crudely estimate VL testing coverage supported by PEPFAR. For example, a comparison may be made between the FY20 Q1 denominator for TX_PVLS and FY19 Q3 TX_CURR, given that patients newly initiating ART and included in TX_CURR in FY19 Q4 and FY20 Q1 may not be eligible for a viral load test. In calculating this estimate, it is important to ensure that individuals, not tests are being reported for TX_PVLS.
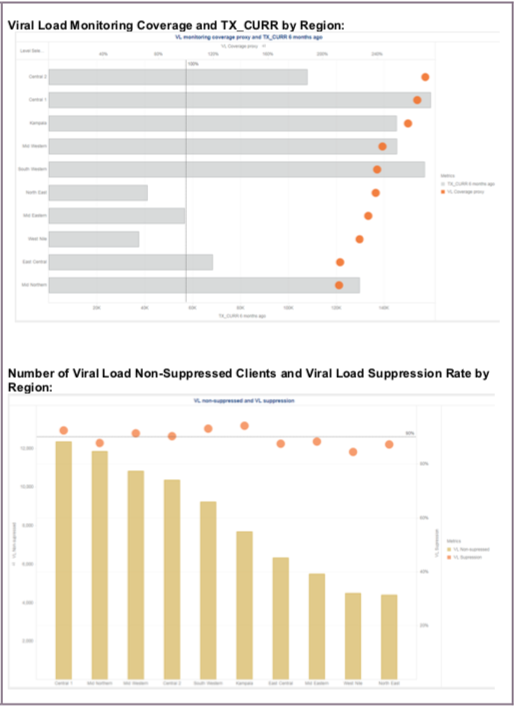
Analyzing both VL testing coverage and suppression rates by geography, sub-population, and implementing mechanisms is essential for program management and quality of care. Real-time review of VL results should trigger an immediate response to follow-up on patients who are not suppressed (i.e., VL ≥1000).
Number of ART patients with suppressed VL results (<1,000 copies/ml) documented in the medical or laboratory records/LIS within the past 12 months
Number of ART patients with a VL result documented in the medical or laboratory records/LIS within the past 12 months.
How to calculate annual total: This is a snapshot indicator. Results are cumulative at each reporting period.
How to collect:
This indicator should be collected from clinical sources (e.g., electronic or paper patient records), where possible, to ensure de-duplicated patient counting and receipt of results to inform patient care. Ideally, data for this indicator should be collected from an electronic medical records system (EMR) to minimize data collection errors and ensure that results are informing patient care. If data collection from an EMR is not possible, indicator data may be collected from paper-based registers or reports that reflect the VL results. If standard patient registers do not contain all the required information, individual patient records should be reviewed.
If a clinical source does not exist or does not contain the desired information, data may be extracted from an electronic laboratory information system (LIS). VL results from an LIS must be linked to back to the individual patients and their record at sites.
NOTE: If patient-linked VL results from LIS is used for reporting, it is incumbent that the implementing partner ensure this information is transcribed into the patient record for timely VL results utilization/patient management.
The data source used for reporting on this indicator should be specified and data reported should be de-duplicated and used to inform patient care at sites. If the LIS is used, please explain why clinical sources could not be used to report on this indicator in the narrative (see guiding narrative question section below).
VL results should be reported for patients who have been on ART for at least 3 months (or according to national guidelines). It is important to ensure that the data sources used to collect and aggregate data are updated to be able to report VL results data for patients who have been on ART for at least 3 months.
Beginning in FY19, this indicator moved from annual to quarterly collection. The reporting period still covers a 12-month period and may include data from the previous fiscal year (see visual below). For example, when reporting data in FY20 Q1, country teams will be required to report data for FY19 Q2+ FY19 Q3+FY19 Q4+ FY20 Q1.

Both only VL tests with recorded results and VL results that are linked back to patients should be included in the numerator and denominator of this indicator. This indicator should be reported for all PEPFAR-supported treatment sites (i.e., from all reporting TX_CURR). VL monitoring result utilization should be promoted for individual patient, site, and program use. If a PEPFAR-supported treatment site (i.e., a site that has reported TX_CURR) has not collected any samples for VL testing, “0” should be entered for both the numerator and denominator.
Where more than one result is available for the reporting period, the most recent result should be reported. Programs should describe the method(s) of data collection and the results de-duplication methodology utilized in their narratives.
Key Populations (KPs):
Both KP-specific and clinical partners should complete these KP disaggregations, but only if safe to maintain these files and to report. Reporting of the key population disaggregation should be consistent with what is described under the KP_PREV “How to review for data quality” section on mutual exclusivity of an individual who falls under multiple KP categories (e.g., FSW who injects drugs). In such instances, the individual should only be reported in ONE KP disaggregation category with which this person is most identified. See Appendix A to support the identification of key populations at service delivery.
The first priority of data collection and reporting of treatment among key populations must be to do no harm. These data must be managed confidentially to ensure the identities of individuals are protected and to prevent further stigma and discrimination of key populations.
Reporting Level: Facility
How to review for data quality:
- Denominator ≥ Numerator: The number of VL results from adults and children on ART must be greater than or equal to the number of VL results from adult and pediatric ART patients with a VL <1,000 copies/ml.
- Numerator ≥ subtotal of each disaggregation: The total number of VL results from adult and pediatric ART patients with a VL <1,000 copies/ml should be greater than or equal to the sum of all of the results disaggregated by age/sex, pregnancy/breastfeeding status, and test indication.
- TX_CURR ≥ TX_PVLS (D): TX_CURR should be greater than or equal to the number of adults and children on ART with VL results
Reporting frequency: Quarterly
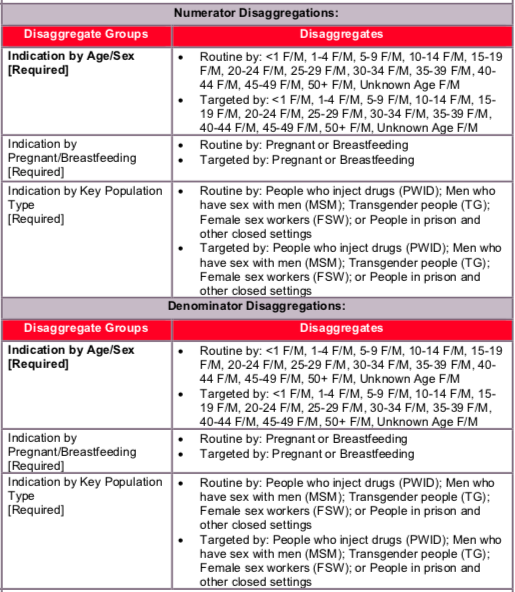
Indication Disaggregate Definitions:
- Routine: Refers to VL tests obtained at standard intervals following ART initiation to monitor virologic response to ART (testing frequencies and interval are dependent on the National guidelines but should be recommended to occur at least annually for patients on ART) and includes follow-up VL tests done after an initial VL result of VL≥1000.
- Targeted: Refers to viral load tests ordered based on a specific clinical indication, (e.g., concern about disease progression or failure to respond to ART).
- If there is more than one VL result for a patient during the past 12 months, report the most recent result.
- Only patients who have been on ART for at least 3 months should be considered.
Only patients who have been on ART for at least 3 months should be considered.
Indicator changes (MER 2.0 v2.3 to v2.4):
- “Not documented” testing indication removed as efforts should have been initiated since this indicator was introduced to move results to either “routine” or “targeted.”
PEPFAR Support definition:
Standard definition of DSD and TA-SDI used.
Provision of key staff or commodities for PLHIV on ART who receive VL monitoring includes: the provision of key staff and/or commodities can include ongoing procurement of critical commodities, such as ARVs, or funding for salaries of HCW who deliver VL monitoring services. Staff who are responsible for the completeness and quality of routine patient records (paper or electronic) can be counted here; however, staff who exclusively fulfill MOH and donor reporting requirements cannot be counted.
Ongoing support for PLHIV receiving ART VL monitoring improvement includes: clinical mentoring and supportive supervision of staff at HIV sites providing ART and VL monitoring services, support for quality improvement activities, patient tracking, enhanced adherence counseling system support, routine support of VL related M&E and reporting, VL related commodities consumption forecasting and supply management.
Guiding narrative questions:
- Briefly describe the VL testing algorithm used in country. Please ensure that the description includes any differences in the VL monitoring algorithm for different sub- populations (e.g., pregnant women, breastfeeding women, children etc.).
- Specify and briefly describe the data sources used to report on this indicator (e.g., EMR, LIS, DHIS 2 etc.). If the LIS is used, please explain why clinical sources could not be used to report on this indicator.
- What efforts are made to ensure individuals, not tests are being reported (e.g., processes of de-duplicating data to reflect unique individuals being tested and outcomes). Please describe the de-duplication methodology used, if applicable.
- Describe the overall coverage of VL testing in the country, with any differences by region or age.
- Describe any association of ART regimen type with TX_PVLS.